May 28, 2026
May 28, 2026
It is pretty heartening to hear Dr Shekhar C. Mande, Director General, CSIR, who said sometime back that CSIR labs are keen to collaborate with anyone who is wanting to work on the m-RNA technology in India (Business Line, 26-1-21).
He further said: “It [m-RNA technology] is an exciting science and this is the first time vaccines” developed by two US companies, Pfizer and Moderna, “have used this technology”. We are willing to partner with anyone who wants to collaborate on this technology”.
These observations are quite encouraging to listen because, amidst the prevailing gloat in print and electronic media about India as a global supplier of vaccines, that someone had at last come forward to talk candidly about our current state of vaccine research and development.
Our extant research capabilities are still confined to age-old technique of producing vaccines with inactivated viruses. Covaxin developed by Biotech, Hyderabad against Covid-19 is one such example. Despite being in the field of vaccine manufacturing since quite some time, that too, with huge production capacities, we have somehow failed to explore new lines of research to master new technologies for developing vaccines. Indeed, m-RNA was advocated as a vaccine platform in the early 1990s. Leveraging this technology, Pfizer and Moderana could launch vaccines against Covid-19 within a short period of identifying the genome structure of the virus. It is against this backdrop that what the CSIR chief said appeals to ears.
That said, we may now take a look at what this m-RNA technology-based vaccine development is all about. To begin with, let us first recall our knowledge about cell biology. As we all know, every normal human cell contains 23 pairs of chromosomes. It means there will be in all 46 chromosomes. They are present in the nucleus of a cell. Some are also present outside nucleus in the mitochondria. Each chromosome contains hundreds to thousands of genes. A trait is a gene-determined characteristic. Some traits are of course, determined by more than one gene. Genes are made up of a chemical known as DNA —deoxyribonucleic acid.
DNA is the hereditary material in humans and almost all other organisms. Each DNA molecule is a long double helix that resembles a spiral staircase. It contains millions of steps. Each step consists of pairs of four types of molecules called bases. In each step, the base adenine (A) is paired with the base thymine (T), or the base guanine (G) is paired with the base cytosine(C). Each base is attached to a sugar (deoxyribose) and phosphate molecules. It is the sugar and phosphate molecules that form the vertical side pieces of the ladder while the base pairs form the ladder’s rungs. Each of this long DNA molecule is coiled up inside one of the chromosomes.
An important property of DNA is: it can replicate. It happens as the entire double-strand molecule splits into two. After splitting, bases on each strand bind to complimentary bases —A with T, and G with C— that are floating nearby. Thus emerges two identical double-strand DNA molecules. Through this process, each new cell forming through cell-division—a process that is essential for cells to multiply and organisms to grow—will have an exact copy of the DNA present in the previous cell.
Proteins, that are essential for the modulation and maintenance of cellular activities, are made out of amino acid building blocks based on the information encoded in DNA. This encoded information in DNA is transferred to messenger ribonucleic acid (m-RNA). m-RNA is synthesized from DNA by an enzyme called RNA polymerase through a process known as transcription.
During the process of transcription, a part of DNA double helix opens and unwinds. One of this unwind strands of DNA acts as template against which a complimentary strand of RNA forms. It is this complimentary strand of RNA that is called messenger RNA (m-RNA). They have a long chain of bases just like a strand of DNA, except that the base uracil (U) replaces the base thymine (T). Unlike DNA, RNAs are usually single stranded. They contain ribose sugars and hence are highly unstable.
Separating from DNA, this messenger RNA, leaving the nucleus travels into the cell cytoplasm and attaches to a ribosome—a tiny structure in the cell where protein synthesis occurs. It then transmits the genetic code that is necessary for protein creation to the ribosome. Decoding the information present in the m-RNA, ribosomes form continuous chains of amino acids to form proteins. Thus, translating the DNA code to ribosome, m-RNA tells it the order and type of amino acids to link together to produce proteins that are needed for our body to remain healthy and alive.
It is on knowing this function of RNA that scientists hypothesised that if we can design our own m-RNA, we can instruct cells (to be precise, ribosomes) to produce any protein that we require. We can create growth agents to mend damaged tissue, say of heart, etc. We can create antibodies to vaccinate against infections. We can even create enzymes to reverse a rare disease.
But while working on this hypothesis, Dr Katalin Kariko, a Hungarian-born biochemist of Pennsylvania University encountered an obstacle. She found that this lab designed m-RNA was found notoriously vulnerable to the body’s natural defences i.e., body sensing that a foreign body intruded, it went to war with it by activating its immune system. In her experiments with animals, she found that synthetic m-RNA generated a massive inflammatory response.
But after years of relentless research, she, in association with Dr Drew Weissman, an immunologist from Penn, discovered a remedy to this vulnerability. In a paper published in 2005, she said that by using a slightly altered nucleoside in the m-RNA string, they could “make it [m-RNA] non-inflammatory and highly translatable” (kariko et al, Immunity 23: 165-175, 2005). At last, she succeeded in reducing body’s immune response to m-RNA, while simultaneously making it efficiently instruct the ribosome to produce the protein that is desired. At the same time, the mRNA code quickly degrades, leaving nothing behind.
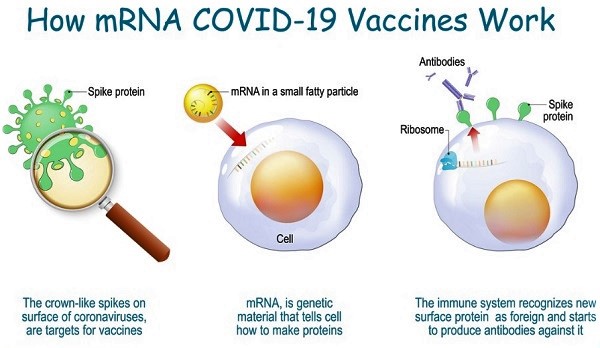
It is this research of Kariko that paved the way for Pfizer-BioNTech combine and Moderna to come up with vaccines against Covid-19 within a short span of time. Unlike traditional vaccines, these m-RNA vaccines instruct our cells to produce a piece of harmless protein similar to that of “spike protein” that is found on the surface of the virus that causes Covid-19.
Thereafter, as the cell displays the protein piece on its surface, our immune system, recognizing it as a foreign body, starts building immune response i.e., makes antibodies, just as it happens when a natural Covid-19 virus attacks us. Having thus learnt to identify the foreign body and make antibodies to fight it out, our bodies keep themselves ready to fight, should a natural covid-19 virus attack us. That is how m-RNA based vaccines give a preview of the virus protein to our bodies and thus help us in protecting ourselves from getting infected if the real corona virus enters our bodies.
So, that’s the story of this “disruptive technology” of vaccine development against infectious diseases. Although there are many unknowns about this technology, scientists do consider it as “cleaner” than conventional vaccine technologies, for the m-RNA code quickly degrades, leaving nothing behind. Secondly, “as any protein can be expressed from mRNA without the need to adjust the production process”, their development, according to Thomas Schlake et al (2012), offers maximum flexibility. Let us therefore, hope that our CSIR laboratories—either alone or in collaboration with private bodies—would catch up with this “game-changing” vaccine technology soon.
Images (c) istock.com
17-Apr-2021
More by : Gollamudi Radha Krishna Murty